
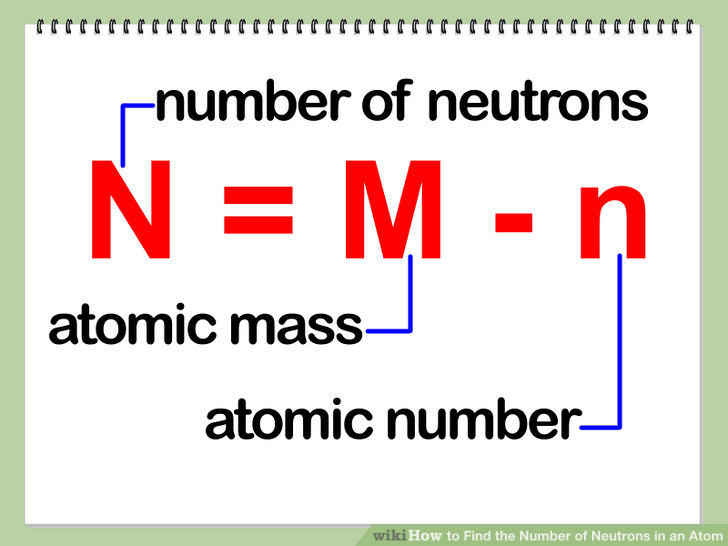
The relative atomic mass is worked out using the following formula, illustrated for two isotopes, where the abundances are given in percentage values. The mass number (A) of an isotope tells us how many protons and neutrons are in the nucleus of an atom of this isotope. In any sample of chlorine, 75 per cent of the atoms are 35 Cl and the remaining 25 per cent are 37 Cl. But the relative atomic mass of chlorine is not 36. The relative atomic mass of an element is a weighted average of the masses of the atoms of the isotopes – because if there is much more of one isotope then that will influence the average mass much more than the less abundant isotope will.įor example, chlorine has two isotopes: 35 Cl and 37 Cl. Calculating relative atomic mass from isotopic abundance 
The A r values also allow you to work out that three oxygen atoms have the same mass as two magnesium atoms.Ĭhlorine’s A r of 35.5 is an average of the masses of the different isotopes of chlorine. They also tell you that hydrogen atoms have 12 times less mass than a carbon atom. These values tell you that a magnesium atom has twice the mass of a carbon atom, and 24 times more mass than a hydrogen atom. M(E) is the atomic mass of the element from the periodic table Example problem: If the masses of one isotope of nitrogen, nitrogen-14, is 14.003 amu and another isotope, nitrogen-15, is 15.000 amu, find the relative abundance of the isotopes.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
